Orbitales hidrogenoides reales
Se representan algunas soluciones reales de la ecuación de Schrödinger para el átomo de hidrógeno.
Estos orbitales representan estados definidos de la energía y del cuadrado del momento
angular. Sin embargo, para m distinto de 0 no son autofunciones del operador L
z.
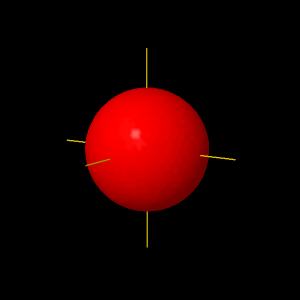
n = 1 , l = 0, m = 0
n = 2 , l = 1, m = 0
n = 2 , l = 1, |m| = 1
n = 3 , l = 2, m = 0
n = 3 , l = 2, |m| = 1
n = 3 , l = 2, |m| = 2
Obsérvese la presencia de nodos en la parte radial de los orbitales 3p.
n = 3 , l = 1, m = 0
n = 3 , l = 1, |m| = 1
n = 4 , l = 3, m = 0
n = 4 , l = 3, |m| = 1
n = 4 , l = 3, |m| = 2
n = 4 , l = 3, |m| = 3
Dpto. de Química Física Aplicada
Based on template by A. Herráez as modified by J. Gutow
Using directory /home/willy/jmol-12.0.19/orbitales-H
adding JmolPopIn.js
...jmolApplet0
...adding 1s.png
...adding 1s.spt
...jmolApplet1
...adding 2pz.png
...copying
/home/willy/jmol-12.0.19/h.log
to
/home/willy/jmol-12.0.19/orbitales-H/h.log
...adding 2pz.spt
...jmolApplet2
...adding 2px.png
...adding 2px.spt
...jmolApplet3
...adding 2py.png
...adding 2py.spt
...jmolApplet4
...adding 3dz2.png
...adding 3dz2.spt
...jmolApplet5
...adding 3dxz.png
...adding 3dxz.spt
...jmolApplet6
...adding 3dyz.png
...adding 3dyz.spt
...jmolApplet7
...adding 3dx2-y2.png
...adding 3dx2-y2.spt
...jmolApplet8
...adding 3dxy.png
...adding 3dxy.spt
...jmolApplet9
...adding 3pz.png
...adding 3pz.spt
...jmolApplet10
...adding 3px.png
...adding 3px.spt
...jmolApplet11
...adding 3py.png
...adding 3py.spt
...jmolApplet12
...adding 4f0.png
...adding 4f0.spt
...jmolApplet13
...adding 4f1.png
...adding 4f1.spt
...jmolApplet14
...adding 4f-1.png
...adding 4f-1.spt
...jmolApplet15
...adding 4f2.png
...adding 4f2.spt
...jmolApplet16
...adding 4f-2.png
...adding 4f-2.spt
...jmolApplet17
...adding 4f3.png
...adding 4f3.spt
...jmolApplet18
...adding 4f-3.png
...adding 4f-3.spt